our approach aimed at maximizing productivity & efficiency.
Founder, Avada Factory Inc.
US FDA Medical Device Classification
The US Food and Drug Administration (FDA) classified medical devices depending on the risk they pose to the user. The FDA divides devices into three classes: Class I (low risk), Class II (moderate risk), and Class III (high risk) (high risk). In addition, the device’s intended function, the location within the patient where it will be utilised, the duration of engagement with the patient, and whether the device is active or passive are all factors that influence FDA medical device classification.
FDA Classification and Regulatory Controls
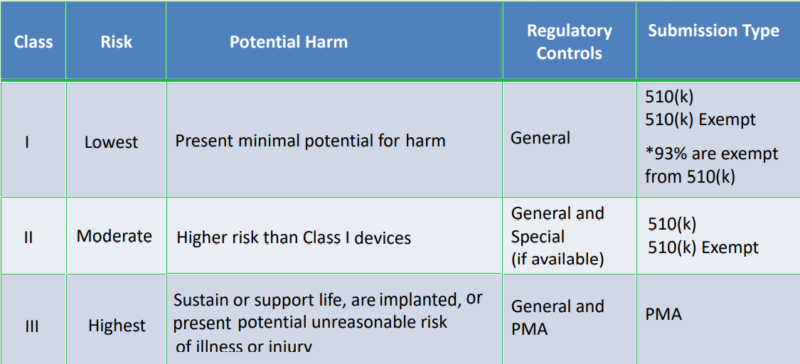
FDA Medical Device Classification Flow Chart